Case captioned: James E. Goetz v. U.S. Food and Drug Administration (Case 3:22-cv-04768-TSH). August 26, 2022 - (MASS TORT NEWS) The Baum Hedlund Aristei & Goldman law firm filed a lawsuit against the U.S. Food and Drug Administration (FDA) last to compel the agency to release documents regarding an agency-sponsored study on Zantac which “has a number of alarming and … [Read more...] about Baum Hedlund Sues FDA to Force Release of Agency Documents Related to ‘Flawed’ Zantac Study
zantac
A Forward-Looking Perspective on Mass Tort Practice
As with most things, the future of mass torts will likely mimic its past. Cancer cases, like those associated with tobacco and asbestos in the past, are driving current and future mass tort litigation. New and ongoing litigation continues for products such as: Zantac Roundup Talc Valsartan Sunscreen PFAS Metformin CPAP With technological and scientific … [Read more...] about A Forward-Looking Perspective on Mass Tort Practice
ZANTAC MDL 2924-Recent Rulings and Overall Status
Including comments by Brent Wisner, Baum Hedlund. MASS TORT NEWS January 18, 2021 Washington, DC In Re: Zantac (Ranitidine) Products Liability Litigation MDL 2924 court website JPML Initial Zantac Transfer Order, Feb. 6, 2020 By Mark A. York, Editor (MassTortNews.org) United States consumers are learning more and more that healthcare manufacturing oversight has failed … [Read more...] about ZANTAC MDL 2924-Recent Rulings and Overall Status
Zantac Litigation Overview
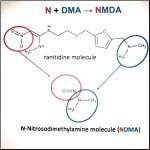
Article Provided by Brenden Leydon Attorney at Wocl Leydon LLC The litigation over the harmful effects of Zantac (generic name is Ranitidine) is just getting underway, and it looks like it will lead to a significant number of cases. What is Zantac/Ranitidine? Ranitidine is an antacid which belongs to a class of drugs known as histamine-2 receptor agonists (H2 blockers). … [Read more...] about Zantac Litigation Overview
FDA Issues Recall Statement On All Zantac (Ranitidine) Products
FDA NEWS RELEASE FDA Requests Removal of All Ranitidine Products (Zantac) from the Market FDA Advises Consumers, Patients and Health Care Professionals After New FDA Studies Show Risk The U.S. Food and Drug Administration today announced it is requesting manufacturers withdraw all prescription and over-the-counter (OTC) ranitidine drugs from the … [Read more...] about FDA Issues Recall Statement On All Zantac (Ranitidine) Products